11 needle-free vaccines that could transform global immunisation
Gavi and its partners have identified 11 needle-free vaccine patches that should be prioritised to boost immunisation coverage in low- and middle-income countries.
- 2 April 2025
- 7 min read
- by Linda Geddes
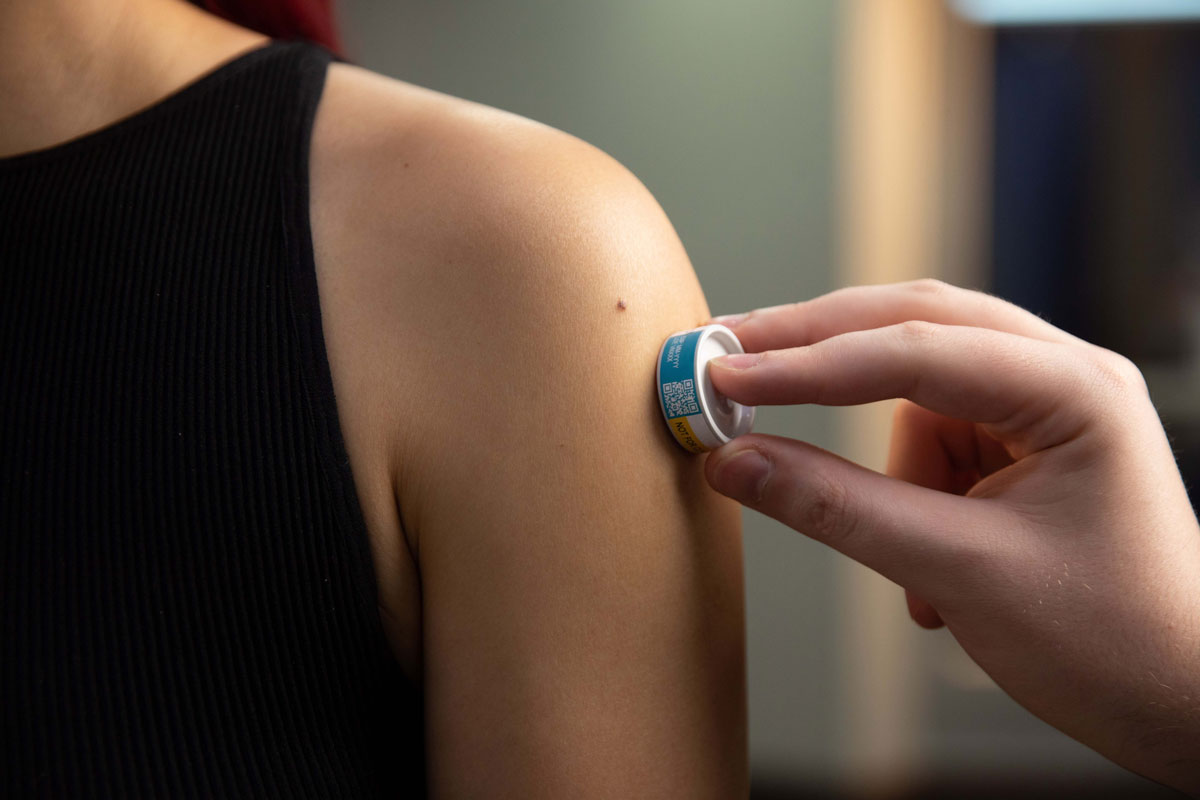
Postable, relatively painless, easier to administer than traditional vaccines, microarray patches (MAPs) could transform immunisation in low-income and pandemic settings.
“Because of these qualities, Gavi and its partners have prioritised MAPs as the number one priority delivery innovation for vaccines, with the potential to increase vaccine coverage and equity in low- and middle-income countries,” says Dr Tiziana Scarna, senior manager in innovation and special products at Gavi, the Vaccine Alliance.
However, a major barrier to developing them is a lack of guidance about which vaccine patches would have the greatest health impact in these settings – and would therefore be most attractive to the governments and organisations who purchase them.
To help address this, the Vaccine Innovation Prioritisation Strategy (VIPS) Alliance – a collaborative effort by Gavi, the World Health Organization (WHO), the Gates Foundation, UNICEF and PATH to evaluate, prioritise and drive forward vaccine product innovations – launched a process to identify which vaccine patches hold the greatest potential to boost immunisation coverage in low- and middle-income countries.
“Development of vaccine MAPs will need a substantial investment from vaccine manufacturers – but manufacturers want to be able to apply this technology to multiple vaccines in their portfolio to reduce the cost of this innovation,” says Dr Birgitte Giersing, team lead for vaccine prioritisation and platforms at WHO, who was involved in the research. “This is why this prioritisation exercise is needed; it signals which vaccine MAPs are considered important and helps de-risk investment decisions.”
Their final assessment, published in Vaccine, identifies 11 candidates that would be most relevant to and valuable for immunisation programmes in low- and middle-income countries.
Here’s what we know about these vaccine candidates and the difference that they could make.
Needle-free immunisation
MAPs are small, sticking plaster-like devices studded with hundreds of microscopic projections that deliver vaccine to the top layers of the skin.
Unlike liquid vaccines, which require constant refrigeration to remain effective, and trained professionals to inject and dispose of the syringes, patches are designed to be more stable at room temperature for a few days, easier to transport, and applied to the skin with minimal training.
Removing the need for needles could also remove one key motivation behind vaccine hesitancy.
“For millions of children in remote, hard-to-reach and conflict-affected communities, MAPs could be a game-changer,” says UNICEF vaccine specialist, Dr Jean-Pierre Amorij, who took part in the research. “These vaccine patches could be a more effective way to reach children who would otherwise miss out on life-saving immunization.”
Indeed, “in some cases, such as for measles, MAPs are considered essential to be able to reach global and regional elimination targets – and are likely to be needed more than ever in the current environment of vaccine hesitancy,” said Giersing.
Because of these attributes, microarray patches have been prioritised by the VIPS Alliance as an innovation that could help address immunisation barriers and increase coverage and equitable access to vaccines. Yet, although the technology underpinning them has been in development for several decades, so far only a handful of vaccine patches have entered clinical trials – including candidates against measles, rubella, COVID-19, seasonal influenza and hepatitis B.
One issue is that microarray patches are considered a novel vaccine product with a complex development pathway. They require significant upfront investment, so manufacturers need to know if there will be a market for these products before committing further resources to developing and testing them.
Prioritisation process
In 2021, the VIPS Alliance published a five-year action plan to help accelerate the development of vaccine microarray patches – including a requirement to identify which patches should be considered the highest priority for lower-income settings.
“There isn’t necessarily a case for replacing needle syringes with patches everywhere. But there are specific cases where we think a patch would be advantageous,” says Scarna, who led the research.
“These are generally vaccines that either need very high population coverage to have the greatest public health impact, those that are not only administered through routine [childhood] immunisation schedules but target other segments of the population for which the immunisation platform is not as well-established, or those that need to be delivered outside of health facilities.”
“It is also important that these products are financially sustainable, so we sought to identify vaccines that could potentially have a market in low- and middle-income, or even high-income countries.”
From an initial reference list of 91 vaccine targets, the VIPS Alliance applied various filters to identify those with the greatest applicability to low- and middle-income countries.
They then consulted an external advisory group to assess the potential regulatory pathways, programmatic impact and financial sustainability of these candidates, before arriving at a final shortlist of 11 vaccine patches.
Priority vaccines
The 11 candidates fell into two categories: priority group one included vaccine targets with high potential programmatic impact, financial sustainability or interest from funders. This included vaccines against hepatitis B virus; measles, mumps and rubella viruses; human papillomavirus (HPV); rabies virus, yellow fever; COVID-19; and both seasonal and pandemic influenza viruses.
Priority group two included vaccines against Group B streptococcus (a leading cause of meningitis and bloodstream infections in newborns); Five strains of Meningococcus bacteria (major causes of neurological disease and disability in sub-Saharan Africa); Salmonella Typhi (which causes typhoid fever); and Pneumococcus (the leading cause of severe pneumonia and bacterial meningitis worldwide).
Of these candidates, measles-rubella vaccine patches are at the most advanced stage of development. A Phase 1/2 study conducted in adults and children in The Gambia found that the vaccine patch was safe and well tolerated, while the immune responses it triggered were similar to those of an injected measles-rubella vaccine.
Micron Biomedical, which developed the measles-rubella vaccine patch, is currently scaling up its manufacturing capacity, ahead of further trials.
“We are hoping that this measles-rubella vaccine will be a trailblazer, and once we will have manufacturing proof of concept for this one, the level of interest and investment in other vaccine microarray patches in the pipeline will increase, accelerating their development,” says Scarna.
Other recent clinical trials that have shown promising results include vaccines against influenza, COVID-19 and hepatitis B.
“An influenza vaccine MAP that eliminates the need for constant refrigeration, improves ease of use and increases a person’s immune response to the vaccine could facilitate administering millions of seasonal influenza vaccines globally every year,” says Collrane Frivold, a Technical Officer in the Medical Devices and Health Technologies Program at PATH, who co-authored the VIPS assessment.
“A hepatitis B vaccine MAP that is easy to use and is more stable at room temperature could improve access to birth-dose vaccination for the millions of newborns who are born outside of health facilities each year, while MAPs for influenza or COVID-19 vaccines could simplify delivery to hard-to-reach populations during future outbreaks.”
Have you read?
Future challenges
Even so, there are many steps that need to occur before such vaccines are ready to be introduced, including late-stage clinical trials, manufacturing scale-up, approvals by national regulatory bodies, WHO prequalification, financing policies and procurement strategies, and implementation research, Frivold says.
“PATH and our VIPS Alliance partners are actively working across these areas to reduce bottlenecks and accelerate MAP introduction timelines for low- and middle-income country markets. Healthcare worker training and advocacy is another essential element of introducing novel vaccine innovations, such as MAPs, to ensure safety, effectiveness, and successful uptake.”
Thorough assessments also provide insights on the potential public health impact and cost effectiveness of the vaccine patches, says Amorij.
“UNICEF has already completed an initial full value assessment of measles and rubella vaccine patches. The findings show that if the patches are broadly deployed, they could reach an additional 80 million children and avert up to 37 million measles cases and 400,000 measles deaths. We now need assessments for other vaccines too, further building momentum around the potential of these patches to protect children from other deadly diseases.”








